
Validated Environmental Monitoring Software for GMP Environments
Compliance built in. Performance built to last
Get a quoteEnvironmental Monitoring Software
Our enVigil software suite is purpose-built for facility and environmental monitoring, developed from over 35 years of experience delivering EU-GMP compliant solutions.
Designed for regulated environments, enVigil provides full oversight of critical parameters including airborne particulates, differential pressure, temperature, humidity, and airflow. It also supports equipment monitoring for fridges, freezers, and incubators, all within a single, validated platform.
From aseptic filling lines and ATMP cleanrooms to hospital pharmacies and IVF clinics, enVigil systems are deployed worldwide to support compliant, real-time monitoring in high-stakes environments.
At its core, enVigil is built around the latest regulatory standards: EU-GMP Annex 1, cGMP, GAMP, and 21 CFR Part 11. Whether you’re upgrading legacy systems or designing a new facility, we help you move towards compliance and stay there with confidence.
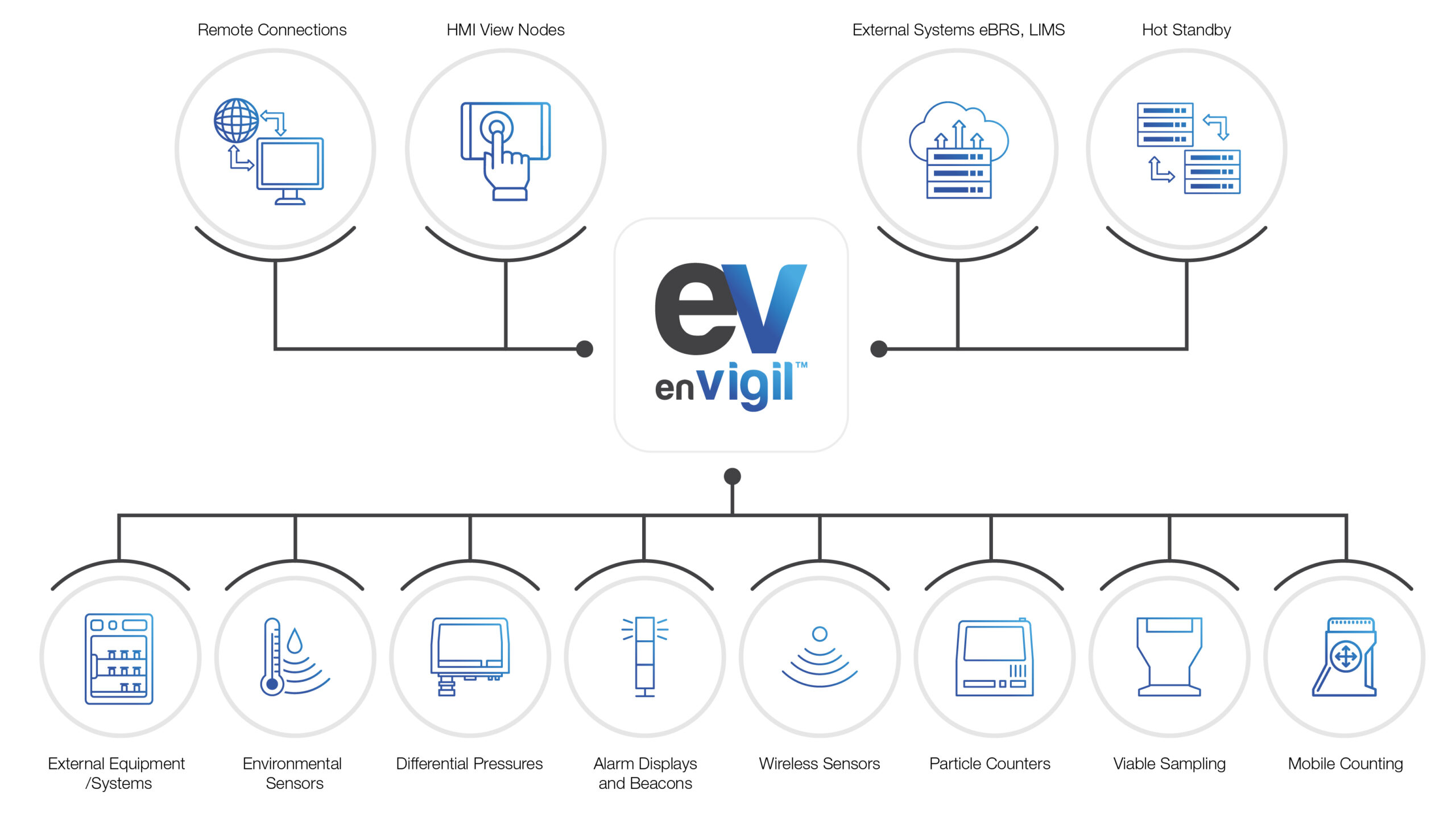
Environmental Monitoring Software for Regulated Facilities
enVigil FMS is designed for organisations that need facility-wide visibility and control across complex, regulated environments.
It is ideally suited to sites where environmental performance must be managed consistently across multiple cleanrooms, controlled spaces and supporting infrastructure, not just monitored in isolation.
By providing a single operational view of environmental conditions across the facility, enVigil FMS helps teams:
- Maintain control across interconnected areas and processes
- Identify trends and deviations early, before they impact compliance
- Coordinate responses across quality, engineering and operations
- Demonstrate ongoing control during audits and inspections
In addition to cleanroom and controlled space monitoring, enVigil FMS supports oversight of critical supporting equipment such as fridges, freezers and incubators, helping ensure environmental consistency beyond primary production areas. The result is greater confidence, reduced operational risk, and clearer accountability across the entire site.
Designed for Cleanrooms, Controlled Areas and Facility Infrastructure
enVigil FMS is deployed across pharmaceutical manufacturing facilities, ATMP cleanrooms, hospital pharmacies and life science environments where site-level environmental control and compliance are essential.
By centralising environmental data across rooms, zones and systems, enVigil FMS enables teams to monitor trends, manage alarms and respond to deviations with confidence.
Built around EU-GMP Annex 1, cGMP, GAMP and 21 CFR Part 11, enVigil FMS supports compliant facility monitoring throughout the lifecycle of the building and its critical infrastructure.
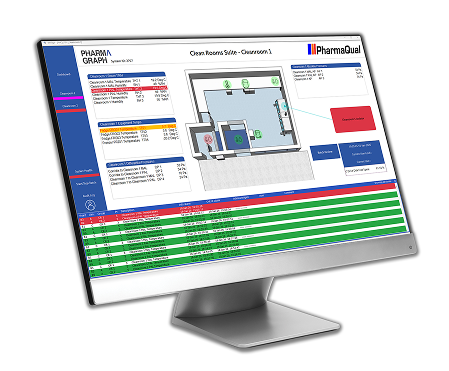

enVigil FMS Environmental Monitoring System
Learn how enVigil FMS supports compliant, site-wide environmental monitoring across regulated facilities, providing consistent oversight of critical environmental conditions.
Download the FMS Brochure Back to ProductsEnvironmental Monitoring Software for Isolator-Based Processes
enVigil IMS is designed to support continuous monitoring at the point of manufacture, where isolators, RABS and aseptic processes demand tight control of environmental conditions.
The system integrates particle monitoring, viable sampling, pressure cascades and environmental parameters into a single process-aware architecture. This allows critical conditions to be monitored in context, not as isolated data points, supporting faster investigation, clearer audit trails and more confident decision-making.

Process Visibility Across the Full Isolator Workflow
Within isolator and filling line environments, enVigil IMS enables monitoring across multiple zones and operational states, supporting normal operation, intervention and recovery phases.
Its modular architecture allows IMS to scale with process complexity, integrating sensors, samplers and alarms in line with defined process requirements, while maintaining a consistent data structure for reporting, trending and review.
Designed to align with EU-GMP Annex 1 expectations for continuous monitoring and data integrity, enVigil IMS supports reliable operation throughout the lifecycle of the isolator or production system.
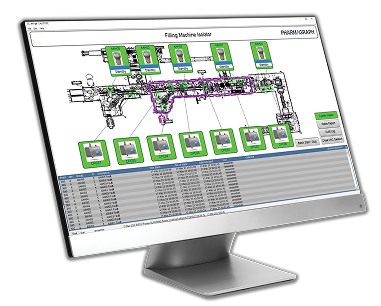

enVigil IMS Environmental Monitoring System
A detailed overview of the features and capabilities of enVigil IMS, our environmental monitoring system designed for process-critical and isolator-based environments, where continuous visibility and data integrity are essential.
Download the IMS Brochure Back to ProductsPre-Configured, Rapid-Deployment Systems
enVigil PnP is a pre-configured environmental monitoring solution designed to simplify deployment, reduce validation effort, and accelerate time to compliance in regulated environments, such as cleanrooms and laboratories.
Built on the proven enVigil platform, enVigil PnP combines validated software, pre-tested hardware and defined system architectures into a ready-to-deploy solution. This approach reduces project complexity while maintaining the robustness required for EU-GMP/GLP regulated applications.
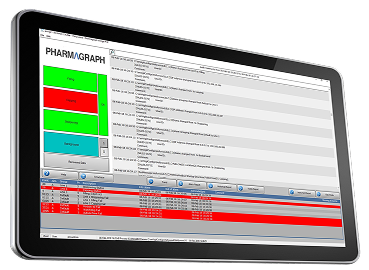
Designed to Reduce Installation and Validation Effort
enVigil PnP is suited to projects where speed, predictability and consistency are critical, including new facility builds, rapid capacity expansion, and repeat installations across multiple sites.
By standardising system configuration and documentation upfront, enVigil PnP helps teams:
- Shorten installation timelines
- Streamline qualification activities
- Reduce engineering and validation risk
- Maintain consistency across deployments
Built around EU-GMP Annex 1, cGMP, GAMP and 21 CFR Part 11, enVigil PnP enables compliant monitoring without the complexity of fully bespoke system design.

enVigil PnP Environmental Monitoring System
A detailed overview of enVigil PnP, a pre-configured environmental monitoring solution designed to simplify deployment and reduce validation effort in regulated environments.
Download the PNP Brochure Back to Products21 CFR Part 11
Compliance Assured
Pharmagraph software is designed to meet the requirements of 21 CFR Part 11, supporting secure data handling, traceability and system integrity in regulated environments.
Key compliance features include:
- Secure user access with unique logins and automatic logout
- Protected binary data storage to prevent record alteration
- Full audit trails capturing what changed, when, by whom and why
- System and operator activity logging, including failed login attempts
Together, these features support faster validation and confident regulatory inspections.


Implementation That Supports Compliance
At Pharmagraph, we understand that compliant software only delivers value when it is implemented correctly within the real operating environment.
That’s why enVigil is supported by experienced project and implementation teams who manage the full delivery lifecycle, from system design and configuration through to installation, validation and handover.
Our teams work closely with quality, engineering and operations stakeholders to ensure monitoring strategies align with regulatory expectations, site workflows and long-term operational needs.
The result is a system that not only meets compliance requirements, but is practical to operate, easy to defend during inspection, and built to support ongoing control.
Accreditations & Assurance
Our pharmaceutical compliance software meets the latest regulatory expectations, including 21 CFR Part 11 and GAMP 5. We build confidence into every system, but we back it with proof.
















